Denmark close to rolling out coronavirus immunity tests: SSI
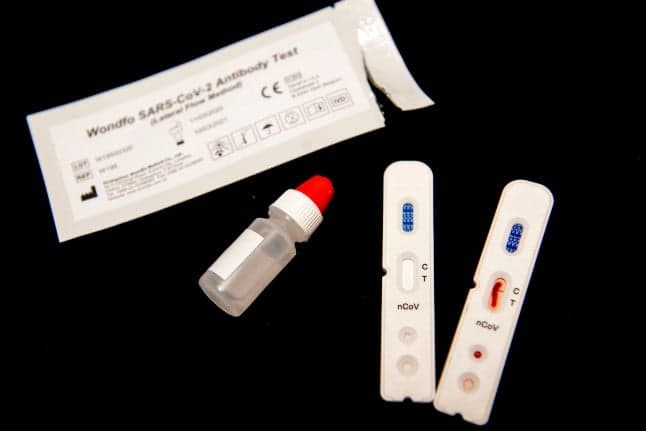
The Danish health agency SSI hopes to soon approve coronavirus antibody tests which will show whether those who suspect they've had the disease are immune and can rejoin the workforce.
“We're not there right now, but we're close," Anders Fomsgaard, the agency's head doctor, told the Politiken newspaper. "I dare not set a date for when the first antibody tests can come out, but my clear expectation is that it will happen before the epidemic culminates.”
Karen Angeliki Krogfelt, professor of medical microbiology at Roskilde University, is working with Statens Serum Institute (SSI) to check the accuracy of the Chinese-made finger-prick blood tests, to determine if they can be used by the Danish authorities.
These so-called 'serologic tests' check for the antibodies produced by the body to defend against the coronavirus virus, rather than testing whether someone is currently infected.
"When people have a high antibody level, they have a very hard time getting infected again for a period of up to two years," Krogfelt told the newspaper. "They can then be sent back to work without much risk of them becoming infected or spreading the infection to others, as they are now more resilient."
Knut Borch-Johnsen, Deputy Director at Holbæk hospital, said that such tests would be invaluable.
"Today, I have to send staff home even if they have very mild symptoms because it might be coronavirus. If we knew they were immune, then some of them could stay at work," he told the newspaper.
The SSI project is being mirrored by governments across the world.
On Monday, Deborah Birx from the White House Coronavirus Task Force said the the US Food and Drug Administration (FDA) hoped to approve a test within weeks, while the UK has already purchased 3.5m tests, and hopes to send them out to people's homes within weeks.
Comments
See Also
“We're not there right now, but we're close," Anders Fomsgaard, the agency's head doctor, told the Politiken newspaper. "I dare not set a date for when the first antibody tests can come out, but my clear expectation is that it will happen before the epidemic culminates.”
Karen Angeliki Krogfelt, professor of medical microbiology at Roskilde University, is working with Statens Serum Institute (SSI) to check the accuracy of the Chinese-made finger-prick blood tests, to determine if they can be used by the Danish authorities.
These so-called 'serologic tests' check for the antibodies produced by the body to defend against the coronavirus virus, rather than testing whether someone is currently infected.
"When people have a high antibody level, they have a very hard time getting infected again for a period of up to two years," Krogfelt told the newspaper. "They can then be sent back to work without much risk of them becoming infected or spreading the infection to others, as they are now more resilient."
Knut Borch-Johnsen, Deputy Director at Holbæk hospital, said that such tests would be invaluable.
"Today, I have to send staff home even if they have very mild symptoms because it might be coronavirus. If we knew they were immune, then some of them could stay at work," he told the newspaper.
The SSI project is being mirrored by governments across the world.
On Monday, Deborah Birx from the White House Coronavirus Task Force said the the US Food and Drug Administration (FDA) hoped to approve a test within weeks, while the UK has already purchased 3.5m tests, and hopes to send them out to people's homes within weeks.
Join the conversation in our comments section below. Share your own views and experience and if you have a question or suggestion for our journalists then email us at [email protected].
Please keep comments civil, constructive and on topic – and make sure to read our terms of use before getting involved.
Please log in here to leave a comment.